Rapid genome sequencing highly effective at diagnosing babies with epilepsy
- Details

Rapid genome sequencing, that can detect rare genetic diseases within weeks, is highly effective at diagnosing babies with epilepsy and leads to better treatment options in most cases, according to a global study.
The research, published in The Lancet Neurology, found rapid genome sequencing had a high diagnostic rate of 43 per cent for infantile epilepsy, supporting the need for greater access to the cutting-edge technology in clinical care.
Infantile epilepsy is a neurological disorder, which causes recurrent and unprovoked seizures, and often leads to serious, lifelong developmental and cognitive delays. Most forms of infantile epilepsy have a genetic cause.
The international study, the first to examine rapid genome sequencing in children with a specific health condition, recruited 100 babies with new-onset epilepsy from Australia, Canada, the UK and the US.
Part of the International Precision Child Health Partnership (IPCHiP), an international consortium, which aims to use genomic data to accelerate discovery and therapeutic development, the Gene-STEPS study was co-led by Murdoch Children's Research Institute, The Hospital for Sick Children (SickKids), Boston Children's Hospital, UCL Great Ormond Street Institute for Child Health and Great Ormond Street Hospital. IPCHiP is the first major global clinical collaboration around genomics and precision child health.
Murdoch Children’s Dr Katherine Howell said early recognition and prompt treatment of infantile epilepsy were crucial to improving health outcomes and preventing further neurological deterioration. The study found 56 per cent of babies who received a diagnosis had their treatment plan changed as result.
“Uncontrolled seizures in a baby’s developing brain can lead to high rates of developmental setbacks,” Dr Howell said. Identifying the cause early can help guide treatment options, such as medication changes, which can improve seizure control. However, current genetic testing methods often take months or even years, resulting in missed opportunities.”
Unlike more targeted genetic testing that is often used to confirm a suspected diagnosis, rapid genome sequencing looks for any changes in a person’s DNA that may explain a medical condition. The technology involves analysing the entire genome, the complete set of an organism's genetic material. Currently, there are more than 800 different genetic causes of infantile epilepsy and many have similar symptoms during infancy.
“We expect rapid genome sequencing to improve the outcomes of babies with epilepsy by providing individualised care, tailored to the underlying cause, at the earliest possible stage,” Dr Howell said. More than half who received a diagnosis in our study saw their treatment change, a remarkable outcome for this vulnerable population.”
But for those whose results did not provide a genetic explanation for their seizures, SickKids Dr Vann Chau said a negative test was also an important piece of information for families.
“Regardless of whether sequencing shows there is a genetic cause, the information gathered from genome sequencing helps clinicians develop a care plan that is specific to the medical needs of each patient,” he said.
Melbourne mum Karsha Trowbridge's son Levi, 3, has KCNQ2 epileptic encephalopathy, a rare genetic disorder that triggers frequent seizures. He was diagnosed as a newborn via rapid genomic testing.

Levi with his parents Karsha and Steve
"I had just bought Levi home from the hospital when he had his first major seizure," she said. It was a terrifying time in the weeks that followed, watching your baby having multiple seizures a day, and at times, turning blue and holding his breath. But when we got a diagnosis that changed everything. We were no longer kept in the dark, not knowing how to help our child.”
Karsha said after changing medications due to the diagnosis, Levi went from having five seizures a day to only a handful a year.
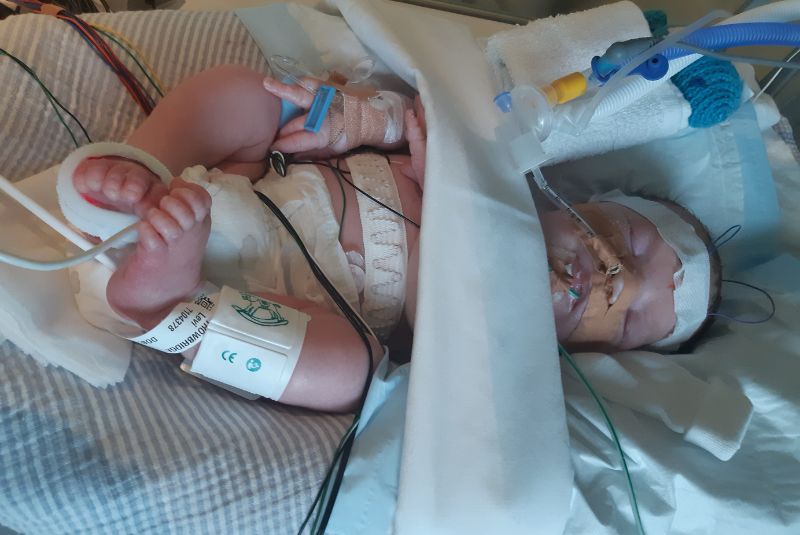
Levi in hospital as an infant
“If we had been waiting years for a diagnosis, his development and learning may never have progressed,” she said. It’s likely he wouldn’t have walked, talked and breathed on his own. Early intervention is crucial because every seizure, no matter how small, does damage.”
Karsha said with the help of various therapies, Levi was a healthy, happy boy whose movement had significantly improved.
“Levi is really kicking goals now,” she said. He can walk, stand in water and pull a part and piece together puzzles.”

Levi playing at the beach
Boston Children’s Hospital Dr Annapurna Poduri said the status quo had been to treat seizures like a symptom and try to find medications that alleviate them. But medications weren’t getting at the underlying causes of epilepsy, she said.
“We all feel a deep sense of responsibility to bring our genetic discovery successes to our patients,” Dr Poduri said. By bringing together experts from across the world in epilepsy and genomics, we demonstrated that if providing an underlying diagnosis to families and providers quickly was made a priority, we could better inform future treatment, evaluation, prognosis, and counselling for families of kids with infantile epilepsy."
SickKids Dr Gregory Costain said thanks to this international, multi-centre partnership, the team was able to demonstrate the feasibility and benefits of performing early rapid genome sequencing across four health-care systems.
“Our hope is that these findings will help us advocate for greater access to genome sequencing in clinical care and lay the groundwork for precision medicine interventions that could one day offer individualised treatment options for each patient and family,” he said.
Great Ormond Street Hospital’s Dr Amy McTague said the study had shown how this testing could be used to diagnose and inform treatment in many children.
“This has the potential to impact many families across the world and provide much needed information to clinical teams in charge of their care,” she said. We are incredibly grateful to every family that took part in this study, research like this is only possible because of them.”
Publication: Alissa M. D’Gama, Sarah Mulhern, MGenCouns, Beth R. Sheidley, Fadil Boodhoo, Sarah Buts, Natalie J. Chandler, Joanna Cobb, Meredith Curtis, Edward J. Higginbotham, Jonathon Holland, Tayyaba Khan, Julia Koh, Nicole S.Y. Liang, Lyndsey McRae, Sarah E. Nesbitt, Brandon T. Oby, Ben Paternoster, Alistair Patton, Graham Rose, Elizabeth Scotchman, Rozalia Valentine, Kimberly Wiltrout, Robin Z. Hayeems, Puneet Jain, Sebastian Lunke, Christian R. Marshall, Shira Rockowitz, Neil Sebire, Zornitza Stark, Susan M. White, Lyn S. Chitty, J. Helen Cross, Ingrid E. Scheffer, Vann Chau, Gregory Costain, Annapurna Poduri, Katherine B. Howell and Amy McTague. ‘Rapid genome sequencing in infantile epilepsy: an international, multicentre, prospective pilot cohort study,’ The Lancet Neurology. DOI: 10.1016/PIIS1474-4422(23)00246-6
*The content of this communication is the sole responsibility of the Murdoch Children’s and does not reflect the views of the NHMRC.
Available for interview
Dr Katherine Howell, Murdoch Children’s, Team Leader, Neuroscience
Karsha Trowbridge whose son Levi, 3, has a rare genetic disorder that causes seizures
Media Contact for Murdoch Children's
Phone:
show phone number
Email:
show email address
About Murdoch Children’s Research Institute
Murdoch Children's Research Institute is the largest child health research institute in Australia committed to making discoveries and developing treatments to improve child and adolescent health in Australia and around the world. They are pioneering new treatments, trialling better vaccines and improving ways of diagnosing and helping sick babies, children and adolescents. It is one of the only research institutes in Australia to offer genetic testing to find answers for families of children with previously undiagnosed conditions.
About The Hospital for Sick Children (SickKids) (Toronto)
The Hospital for Sick Children (SickKids), affiliated with the University of Toronto, is Canada's most research-intensive hospital and the largest centre dedicated to improving children's health in the country. With passionate and dedicated staff, SickKids is much more than a hospital. We work each day to provide the best in child and family-centred care, create ground-breaking clinical and scientific advancements, and train the next generation of experts in child health. SickKids is a founding member of Kids Health Alliance, a network of partners committed to advancing excellence and expertise in the delivery of paediatric care in Ontario.
About Great Ormond Street Hospital and Great Ormond Street Hospital Children’s Charity
Great Ormond Street Hospital is one of the world’s leading children’s hospitals with the broadest range of dedicated, children’s healthcare specialists under one roof in the UK. The hospital’s pioneering research and treatment gives hope to children from across the UK with the rarest, most complex and often life-threatening conditions. Our patients and families are central to everything we do – from the moment they come through the door and for as long as they need us.
Great Ormond Street Hospital Children’s Charity needs to raise money to support the hospital to give seriously ill children, the best chance for life. The charity funds research into pioneering new treatments for children, provides the most up to date medical equipment, funds support services for children and their families and supports the essential rebuilding and refurbishment of the hospital. You can help us to provide world class care for our patients and families.
About Boston Children’s Hospital
Boston Children’s Hospital is ranked among the best children’s hospitals in the nation by U.S. News & World Report and is a paediatric teaching affiliate of Harvard Medical School. Home to the world’s largest research enterprise based at a paediatric medical centre, its discoveries have benefited both children and adults since 1869. Today, 3,000 researchers and scientific staff, including 11 members of the National Academy of Sciences, 23 members of the National Academy of Medicine and 9 Howard Hughes Medical Investigators comprise Boston Children’s research community. Founded as a 20-bed hospital for children, Boston Children’s is now a 485-bed comprehensive centre for paediatric and adolescent health care.
Funding
The BCH site is supported by the BCH Children’s Rare Disease Cohorts Initiative and the One8 Foundation. AMD is supported by the National Institute of Child Health and Human Development (T32 HD 098061) and the American Academy of Pediatrics (Marshall Klaus Neonatal-Perinatal Research Award). BRS, RV and APoduri are supported by the One8 Foundation and BRS, JK, BTO and APoduri are supported by the Robinson Family Initiative for Transformational Research. The Toronto authors received support from the Canadian Institutes of Health Research (Funding Reference Number: PJT186240); Epilepsy Canada (New Investigator Award to GC); Feiga Bresver Academic Foundation; Ontario Brain Institute (This research was conducted with the support of the Ontario Brain Institute, an independent non-profit corporation, funded partially by the Ontario government. The opinions, results and conclusions are those of the authors and no endorsement by the Ontario Brain Institute is intended or should be inferred) and University of Toronto McLaughlin Centre. RZH is supported by a Canada Research Chair in Genomics and Health Policy (CRC-2021-00284). Funding for the MCC site was provided by The Royal Children's Hospital Foundation and seed funding from Murdoch Children’s. Murdoch Children’s is supported by the Victorian Government's Operational Infrastructure Support Program. KBH is supported by a Melbourne Children's Clinician Scientist Fellowship. IES and KBH are supported by the National Health and Medical Research Council (NHMRC) Centre for Research Excellence Grant (GNT2006841). IES has a NHMRC Synergy Grant (GNT2010562) and Senior Investigator Grant (GNT1172897) and is supported by the Australia Medical Research Future Fund and Australian Epilepsy Research Fund. AM is supported by the MRC (MR/T007087/1), GOSH Charity (VS0122), Rosetrees Trust and Wellcome Trust TIN Scheme. SEN, ES, LSC, JHC, and AM are supported by the GOSH NIHR Biomedical Research Centre (BRC). LSC is supported by the GOSH Children’s Charity and received discounted consumables from Illumina, Inc. JH is supported by an Action Medical Research and British Paediatric Neurology Association Research Training Fellowship (GN2945). This work is partly funded by the NIHR GOSH BRC. The views expressed are those of the authors and not necessarily those of the NHS, the NIHR, or the Department of Health.


